- #ElectionsWithNewsGram
- HOME
- OPINION
- ON GROUND
- INTERVIEW
- IPL 2026
- INDIA
- NewsGram USA
- WORLD
- न्यूजग्राम
- POLITICS
- ENTERTAINMENT
- CULTURE
- LIFESTYLE
- ECONOMY
- SPORTS
- Jobs / Internships
- Misc.
- NewsGram Exclusive

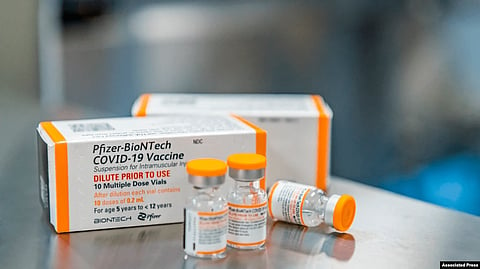
The U.S. Food and Drug Administration authorized on Friday the Pfizer-BioNTech COVID-19 vaccine for emergency use in children 5-11 years old.
The FDA approved for children doses that are one-third the amount that teens and adults receive.
"With this vaccine kids can go back to something that's better than being locked at home on remote schooling, not being able to see their friends," Dr. Kawsar Talaat of Johns Hopkins University said, according to The Associated Press. "The vaccine will protect them and also protect our communities."
On Tuesday, advisers to the Centers for Disease Control and Prevention will make detailed recommendations, and the CDC director will have the final say.
Approval by the regulatory agencies would make the vaccine available in the coming days to 28 million American children, many of whom are back in school for in-person learning. Only a few other countries, including China, Cuba and the United Arab Emirates, have so far cleared COVID-19 vaccines for children in this age group and younger.
A youth receives the Pfizer-BioNTech Covid-19 coronavirus vaccine at Cu Chi Town Primary School in Ho Chi Minh City on Oct. 27, 2021, as Vietnam began vaccinating adolescents between the ages of 12 to 17. Image credit: VOA
Meanwhile, the World Health Organization Regional Office for Europe on Friday called for schools to stay open, provided appropriate prevention and response measures are in place.
The recommendation comes after WHO reported the European region has now seen four consecutive weeks of growing COVID-19 transmission, the only WHO region to do so. The agency said Europe's rising numbers accounted for 57% of new cases worldwide in the third week of October.
In a statement from the agency's website, WHO/Europe says instead of closing educational institutions in response to this latest surge, it recommends a "whole-of-society approach" to reducing transmission through mitigation measures such as maintaining physical distancing, cleaning hands frequently, wearing masks and ensuring adequate ventilation.
The WHO regional director for Europe, Dr. Hans Henri Kluge, said, "Last year's widespread school closures, disrupting the education of millions of children and adolescents, did more harm than good, especially to children's mental and social well-being. We can't repeat the same mistakes."
Kluge said that in the coming months, decisions by governments and the public to reduce the impact of COVID-19 should be based on data and evidence, "with the understanding that the epidemiological situation can change, and that our behavior must change with it. Science must trump politics." (VOA/RN)
Keywords: FDA, Pfizer, Vaccine, COVID-19
Subscribe to our channels on YouTube and WhatsApp
Download our app on Play Store
